Abstract
Objectives: To evaluate the prevalence and the factors associated with recurrence of myasthenia gravis following thymectomy.
Methods: Six electronic databases which reported on recurrence of myasthenia gravis following thymectomy and/or its risk factors from 1985 to 2018 were searched. Summary prevalence and risk values obtained based on the random effect models were reported.
Results: Seventy (70) papers containing 7,287 individuals with myasthenia gravis who received thymectomy as part of their management were retrieved. The patients had a mean follow-up of 4.65 years post-thymectomy. The prevalence of myasthenia gravis recurrence post-thymectomy was 18.0% (95% CI 14.7–22.0%; 1865/7287). Evident heterogeneity was observed (I2=93.6%; p<0.001). Recurrence rate was insignificantly higher in male compared with female patients (31.3 vs. 23.8%; p=0.104). Pooled recurrence rates for thymomatous (33.3%) was higher than the rate among non-thymomatous (20.8%) myasthenia gravis patients (Q=4.19, p=0.041). Risk factors for recurrence include older age, male sex, disease severity, having thymomatous myasthenia gravis, longer duration of the myasthenia gravis before surgery, and having an ectopic thymic tissue.
Conclusion: A fifth of individuals with myasthenia gravis experience recurrence after thymectomy. Closer monitoring should be given to at-risk patients and further studies are needed to understand interventions to address these risks.
Myasthenia gravis (MG) a rare clinical condition characterized by autoimmune abnormalities is also the commonest neuromuscular junction (NMJ) disorder.1 Its pathophysiology involves the production of abnormal antibodies which binds to nicotinic acetylcholine receptors at the NMJ of skeletal muscles leading to alteration and damage of the NMJ.1-2 The disease is common in young women, however, another peak in incidence may occur at the 6th or 7th decade of life mainly in men in some population; and it may exhibits no sex preference in others.2 Clinically, patients with MG develop varying levels of skeletal muscle weakness affecting the ocular, bulbar, oculo-bulbar system, respiratory system, and the extremities.1 The pattern and course of MG is complex, varying from early remission to acute exacerbation and even death.3
Previous research has revealed a strong relation between MG and disorders affecting the thymus. About 40 to 70% of individuals with MG have thymic follicular hyperplasia, and 10 to 21% of them have thymoma.4-6 Also, 20-47% of individuals having a thymoma have already developed or will develop MG.6-7 Findings from the literature indicate that 40 to 90% of individuals with MG attained remission following thymectomy compared with 10 to 20% among individuals with MG treated with medications without any surgery.6,8 Also, a recent randomised controlled trial demonstrated that thymectomy for individuals with nonthymomatous MG demonstrated better treatment outcomes during a three-year period compared with pharmacological therapy alone.9 Thus, guidelines now recommend thymectomy as a key treatment approach for MG.10
A number of systematic reviews and/or meta-analyses among individuals with MG have been carried out. Some of these reviews explored the differences in outcomes between thymectomy and conservative management of MG,11,12 others compared surgical approaches of thymectomy,13-15 or potential prognostic factors for remission among individuals with MG irrespective of histological type following thymectomy.3,16 None assessed recurrence of MG following thymectomy or its risk factors. The identification of patient factors which are associated with recurrence of MG following thymectomy is crucial for the development of targeted interventions to address challenges associated with the care of individuals with MG. This systematic review and meta-analysis aimed to estimate the post-thymectomy recurrence rates and to investigate the factors associated with recurrence of MG following thymectomy in individuals with MG.
Methods
This systematic review was conducted according to PRISMA guidelines.17 We performed an electronic search of: CINAHL, Clarivate Analytics Web of Science, Cochrane Library, SCOPUS, MEDLINE and DARE databases for relevant articles published between January 1985 and September 2018 which reported on recurrence of myasthenia gravis and its risk factors post-thymectomy. Keywords related to the study were used to evaluate the titles or abstracts of the papers identified during the literature search. Both terms retrieved from Medical Subject Heading (MESH) as well as Boolean operators were utilized during the search. We limited the literature search to publications in English. Search terms used included: [[“Thymus”] OR [“Thymoma”] OR [“Thymectomy”] OR Thym.ti,ab.] AND [[“Myasthenia gravis”[MeSH]] AND [[“Myasthenia”[All Fields] AND “gravis”[All Fields]] AND [[“recurrence”[MeSH] OR “recurrence”[All Fields] OR [“Worsening” OR “Remission”] OR [“risk factors” OR “determinants” OR “predictors”]. Furthermore, the references of published original studies and review papers were assessed for other potentially-relevant publications.
Inclusion and exclusion criteria
All publications on thymectomy in individuals with MG over the given period were included. The criteria for eligibility or inclusion is meeting the following conditions: (i) the population evaluated in the study consisted of mainly adults i.e., 18 years old or older; (ii) the publication reported on outcomes of thymectomy in individuals with MG, (iii) the outcome included recurrence defined based on the Myasthenia Gravis Foundation of America: as worsening, exacerbation or unchanged clinical status of the MG following thymectomy;18 (iv) Individuals with MG who had thymectomy were monitored (followed-up) for at least six months or more after the surgical procedure; and (vi) regression analysis was performed to identify factors associated with the outcomes of the MG.
Publications with the following characteristics were excluded: (i) the age group of the population evaluated was not defined; (ii) publications that included individuals with MG who did not receive thymectomy as a management approach; (iii) studies that report post-thymectomy myasthenic crisis that occurred during or the immediate post-operative period, as well as immediate post-operative outcomes of thymectomy in individuals with MG, (iv) Studies on post-thymectomy MG which developed in individuals with thymoma who had no preceding presentation of myasthenia gravis, (v) Studies with a sample size less than 10. Also, case reports, conference abstracts, editorials, commentaries, review articles and duplicate publications were deemed ineligible.
Data extraction
Two reviewers (FA and SA) independently reviewed the title and abstract of the identified studies. All disagreements between the reviewers were handled by discussion and reaching a consensus. The complete articles of papers identified for detailed review and data extraction were obtained. The 2 reviewers (FA and SA) performed the data extraction independently. Any conflicts between them was also reconciled by discussion and reaching a consensus. The information was retrieved from each of the selected studies include: author and year, country of study, date of data collection, number of patients, recurrence rates, and duration of monitoring or follow-up after the surgical procedure. The selected studies quality were extensively evaluated with the Joanna Briggs Institute’s prevalence critical appraisal checklist for the evaluation of the quality of relevant publications.11-19
Statistical analysis
Summary prevalence values obtained based on the random effect models with their 95% confidence intervals (95% CIs) were reported.20 The Q and I2 statistics were used to assess for the occurrence of heterogeneity between the studies.21 Publication bias was evaluated using an assessment of the funnel plot symmetry and the Egger’s regression test. Also, the effect size measures used were extracted from the selected studies. The odds ratios (ORs) and their 95% CIs were extracted or where not available, were estimated (using the proportion of recurrence of MG post-thymectomy (ie, recurrence cases]) and non-recurrence of MG (ie, non-recurrence [controls]) groups) for each risk factor. All analyses were performed using thee Comprehensive Meta-Analysis software 2.2 (Biostat Inc, USA).22 Sub-group meta-analysis was performed for the following groups: sex (male versus female), and type of myasthenia gravis (thymomatous versus non-thymomatous).
Results
Characteristics of the studies
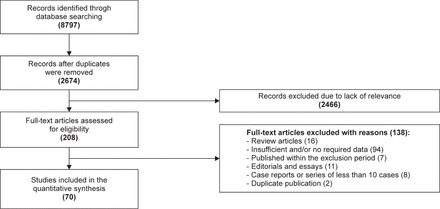
The systematic search of the literature identified 8,797 potentially-relevant articles (Figure 1). Only 2,674 relevant articles remained after duplicate publications were identified and removed. Also, further analysis of the abstracts of the papers led to the removal of another 2,466 articles for lacking relevance to the study objectives. A total of 208 articles remained eligible for full text assessment. Overall, a total of 70 papers from 27 countries published between January 1985 and September 2018 met the inclusion requirements and were selected.6,23-91 Table 1 summarizes the characteristics of the studies included in the review. Majority of the selected publications 52 (74.2%) reported data on both thymomatous and non-thymomatous MG, 15 (21.4%) of the studies reported data only on non-thymomatous MG and 3 (4.3%) reported data only on thymomatous MG. Overall, 7,287 individuals with MG were analyzed in the review. The follow-up time of the individuals with MG following thymectomy ranges from 0.5 to 13.3 years, (mean: 4.65 years).
Flow chart for article identification and exclusion process.
Characteristics of studies included in the systematic review.
Quality assessment of included studies
The quality of the studies was evaluated according to 10 domains. The included papers had good quality in ≥5 of the domains assessed. Majority of the articles (n=36/70) met the requirements for high quality papers (a score of between 7 and 10 of the quality domains evaluated), and others (n=34/70) met the requirements for moderate quality papers (a score of between 5 and 6 in the quality domains evaluated). The commonest quality domains not met by the publications were: having small sample size, low-quality statistical analytical approach, lack of adjustment for confounders, and absence of sub-group analysis.
Prevalence of myasthenia gravis recurrence post-thymectomy
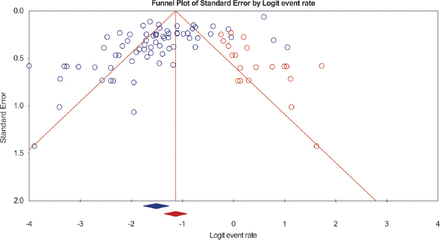
Table 2 shows the summary analysis of the rate of recurrence of MG following thymectomy. The prevalence of MG recurrence post-thymectomy was 18.0% (95% CI 14.7–22.0%; 1865/7287). However, there was heterogeneity between the studies (I2=93.6%; p<0.001). Also, the assessment of the funnel plot (Figure 2) indicate that there may be publication bias (p<0.001). We also explored sex differences in the recurrence rates of MG post-thymectomy. Overall, 13 studies reported data on the recurrence rates of MG post-thymectomy disaggregated according to sex of the patients. There was a high prevalence of recurrence of MG post-thymectomy in males compared with females (31.3 vs. 23.8%) respectively; but the differences between group was not significant (Q=2.60; p=0.107). In addition, studies that reported disaggregated data for thymomatous and non-thymomatous MG showed that pooled recurrence rates for thymomatous MG (33.3%) were higher than the rates among non-thymomatous MG (20.8%); and the difference was statistically significant (Q=4.19, p=0.041).
Pooled event rates of myasthenia Gravis recurrence Post-thymectomy.
Forest plot of the meta-analysis on prevalence of myasthenia gravis recurrence post-thymectomy. CI - confidence interval
Risk factors for recurrence of myasthenia gravis post-thymectomy
Table 3 summarises the meta-analysis of common risk factors of recurrence of MG post-thymectomy. A meta-analysis of eight studies showed that older age (>35–60 years) was a risk factor of recurrence (OR 3.5; 95% C.I. 2.4–5.0). No heterogeneity was found between the studies (I2=0), but evidence of publication bias existed (p=0.017). In a meta-analysis of five studies, male sex was also a risk factor for recurrence of MG following thymectomy (OR 2.8; 95% C.I. 1.5–5.4). There was minimal heterogeneity among the studies (I2=34.8) as well as some publication bias (p-value <0.001). Also, a higher MG stage was a risk factor of recurrence following thymectomy in a meta-analysis of 10 studies (OR 4.8; 95% C.I. 3.1–7.7). There was minimal heterogeneity among the studies (I2=39.6) and some publication bias (p=0.03).
Summary of meta-analysis of risk factors of myasthenia gravis recurrence post-thymectomy.
Furthermore, a meta-analysis involving nine studies revealed that having a thymomatous MG was a risk factor for recurrence post-thymectomy (OR 2.1; 95% C.I. 1.6–2.7). There was minimal heterogeneity (I2=24.3), but no publication bias was noted among the studies (p=0.118). A meta-analysis of six studies found that longer duration of the MG prior to the thymectomy was a risk factor for recurrence post-thymectomy (OR 2.3; 95% C.I. 1.04–4.9). A high level of heterogeneity was noted among the studies (I2=79.2), but publication bias was not evident (p=0.433). In addition, having an ectopic thymic tissue was a risk factor of MG recurrence post-thymectomy according to a meta-analysis of three studies (OR 6.04; 95% C.I. 3.0–11.82). There was absent heterogeneity (I2=0) and publication bias between the studies (p=0.687). A meta-analysis of four studies revealed that having a thymic hyperplasia was not a risk factor for recurrence of MG post-thymectomy (OR 1.1; 95% C.I. 0.2–757).
Thymic atrophy (OR 2.5; 95% C.I. 1.4–4.6), higher dosage of pyridostigmine (OR 2.8; 95% C.I. 1.4–5.3), and use of steroid before thymectomy (OR 2.6; 95% C.I. 1.5–4.5) were all found to be potential risk factors of MG recurrence post-thymectomy in a meta-analysis of two studies each (Table 3); however, the inadequate number of studies precluded any further analysis of these factors. Other less common risk factors for MG recurrence following thymectomy are summarised in Table 4. Overall, a duration of less than six years post-surgery (OR=1.37), having a previous thymectomy (OR=4.6), receiving plasampheresis post-surgery (OR=3.4), hyperthyroidism (HR=8.3), preoperative myasthenic crisis (OR=4.7), having bulbar symptoms (OR=3.3), long duration of thymectomy procedure (OR=1.03) and higher blood loss during surgery (OR=1.02) were all found to be potential risk factors.
Less common risk factors of myasthenia gravis recurrence post-thymectomy.
Discussion
This systematic review of observational studies demonstrated that 18.0% of patients who had thymectomy as part of their management for MG experienced recurrence during follow-up. Also, post-thymectomy MG recurrence rates were insignificantly higher in males compared with females and was significantly higher in individuals having thymomatous than non-thymomatous MG. In addition, we found that significant risk factors for MG recurrence post-thymectomy include: older age, male sex, MG severity, increased MG disease duration pre-surgery and ectopic thymic tissue. Other potential risk factors for MG recurrence post-thymectomy were also identified.
Previous meta-analyses were carried out either to evaluate the effectiveness of thymectomy relative to pharmacological therapy or to assess the effectiveness of thymectomy to achieve complete stable remission.11,14-16 This is the first meta-analysis that assessed the recurrence rates of MG in patients who have had thymectomy. Our analyses showed that almost a fifth of the patients experienced MG recurrence after a mean follow-up duration of about 4.7 years post-thymectomy. This finding further lends credence to a recent report of a randomised controlled trial which demonstrated the additional benefit of surgical treatment (thymectomy) relative to pharmacological therapy alone for the management of individuals with MG.9 This indicates that thymectomy is an important management strategy but additional care needs to be given in order to prevent recurrence among affected patients.
We found that older patients were over three times more likely to experience recurrence of their MG following thymectomy. Previous studies showed that younger age at onset of MG (<40 years) was a good prognostic factor for remission among thymectomized patients while others did not demonstrate this finding.11,30,92 Our analysis demonstrates that older age is a risk factor for MG recurrence. The reason for the effect of age on recurrence rates among the patients is not clear. It may be that younger patients are more clinically-stable to tolerate thymectomy compared to older individuals. There is a need for properly-designed analytical studies to further evaluate the effect of age in individuals with MG who had had thymic excision. Also, sex is a biological factor showing variable associations in studies of individuals with MG. Some literature have reported improved outcomes in female subjects while others either reported improvements in males or no sex differences.27,75 Our analysis indicates that men were almost thrice as likely to experience recurrence of MG following thymectomy than women. There is therefore a need for closer monitoring of men with MG during treatment.
Furthermore, higher stage of the MG (disease severity) was a risk factor of MG recurrence post-thymectomy. Previous studies have demonstrated that MG patients with mild disease had substantially increased survival and remission rates compared to individuals having severe MG.27,93 However, our finding may partly be because individuals with MG who underwent thymectomy were more likely to have had a more severe disease than those who did not.94,95
Also, we found that disease duration before surgery was a risk factor for recurrence. Previous studies have demonstrated that a longer duration of MG before the surgery is a prognostic factor for remission.96,97 The reason why patients with longer duration of MG have higher rates of recurrence deserves further evaluation.
Thymic histology has been found to be a relatively consistent determinant of treatment response in individuals with MG. In many studies, hyperplasia has been found to be related with higher improvement rates.27,77,98 Our meta-analysis demonstrates that hyperplasia was not a predictor of MG recurrence following thymectomy. However, patients having an ectopic thymic tissue were 6 times more likely to experience recurrence following MG. This indicates that patients with ectopic thymic tissue should be given closer monitoring and care following thymectomy.
A number of factors like thymic atrophy, higher pyridostigmine dosage (>240 mg) and use of steroid before thymectomy were all found to predict a higher risk of MG recurrence following thymectomy. A number of other potential risk factors found in single studies were: less than 6 years post-surgery, previous thymectomy, receiving plasmapheresis post-surgery, hyperthyroidism, preoperative myasthenia gravis crisis, having bulbar symptoms, longer operation time and higher blood loss during surgery. These factors need to be explored further in future studies.
There are a number of strengths and limitations of this analysis. A major strength is that we evaluated recurrence rates in 7,287 patients with MG following thymectomy. Second, although the studies included used varying classification systems and reporting of crude rates of remission and improvement, we were able to group the selected studies according to recurrence rates. Third, we identified major risk factors for MG recurrence following thymectomy. However, there are some limitations that require consideration. First, our meta-analysis mainly included descriptive and analytical studies; therefore, differences in the study design, patient characteristics and the type of care given before surgery may introduce some heterogeneity among the patients. Second, most of the studies included in the analyses have relatively small sample size and the risk factors obtained were not controlled for confounders. It may be likely that the pooled risk factor groups identified might overestimate the risk. Other sources of bias may be differences in the surgical skills of the surgeon who performed the thymectomy and the surgical approach used. Despite these limitations, the study provides valuable data necessary for clinical care and policy.
In conclusion, there is increasing interest on the effect of thymectomy in the management of MG and a better understanding of recurrence rates following thymectomy and its prognosis are crucial for clinical practice. We found that a fifth of the MG patients who had thymectomy developed recurrence and a number of demographic and clinical factors are predictors of MG recurrence post-thymectomy. Closer attention should be given to MG patients with the identified risk factors and there is a need for further well-designed studies to assess the effect of confounding on the risk factors identifies and a closer assessment of emerging risk factors.
Copyright
Whenever a manuscript contains material (tables, figures, etc.) which is protected by copyright (previously published), it is the obligation of the author to obtain written permission from the holder of the copyright (usually the publisher) to reproduce the material in Saudi Medical Journal. This also applies if the material is the authors own work. Please submit copies of the material from the source in which it was first published.
Acknowledgment
We would like to thank the professional editors at Editage, a division of Cactus Communications for English language editing.
Footnotes
Disclosure. Authors have no conflict of interests, and the work was not supported or funded by any drug company.
- Received January 31, 2020.
- Accepted August 30, 2020.
- Copyright: © Neurosciences
Neurosciences is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.