Abstract
Objective: To retrospectively compare 2 injection techniques in the management of spastic equinovarus deformity after stroke.
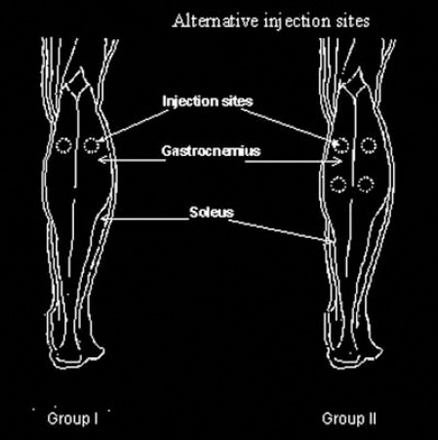
Methods: Patients with stroke were seen at King Hussein Medical Center, Amman, Jordan between January and December 2009. The study design involved an open label retrospective analysis of medical records of 2 groups of comparable age and onset of first stroke. Botulinum toxin was injected into the calf muscles at 2 sites in group I (12 patients) and 4 sites in group II (14 patients). Functional gain was evaluated by the time to walk 10 meters at month one, 3, and 6 compared with baseline.
Results: There was significant improvement in walking time in each study group. However, there was no significant difference between the 2 groups as measured by the 10-meter walking time.
Conclusion: Fewer injection sites would minimize patient discomfort and possibly the production of antibodies, yielding similar therapeutic effects.
Stroke is the second leading cause of death and disability globally, according to the World Health Organization.1 More than one third of stroke survivors never regain adequate limb function.2 A contributing impediment to regaining function is spasticity. Defined as a motor disorder characterized by a velocity-dependent increase in tonic stretch reflexes (muscle tone) with exaggerated tendon jerks, spasticity results from hyper-excitability of the stretch reflex, one component of upper motor neuron syndrome.3 This may affect limb position and activities of daily living.4 Spasticity contributes to disability in post-stroke patients with a frequency of 41.6%.5 Botulinum toxin is a neurotoxin that acts on the neuromuscular junction at the presynaptic site. It inhibits the release of acetylcholine, thus leading to muscle weakness and reduced muscle tone.6 The use of botulinum toxin type-A (BTX-A)7,8 in the management of spastic equinovarus deformity after stroke has been shown to be effective in reducing muscle tone, reducing pain, improving gait, and facilitating other rehabilitation programs.9,10 Side effects of botulinum toxin include local skin reaction and the spread of weakness to the adjacent non-injected muscles. Contraindications to botulinum toxin include pregnancy, lactation, neuromuscular junction disorders, and concurrent use of aminoglycosides.11 However, so far there has been no agreement on the number of injection sites for the best delivery of the toxin to produce the maximum therapeutic effects.12,13 The objective of the current study was to compare the clinical efficacy of injecting BTX-A (Dysport®) into 2 sites (the lateral and medial heads of gastrocnemius) with a 4-site technique (gastrocnemius and soleus) in the management of post-stroke lower limb spasticity.
Methods
This study was part of a Clinical Audit Program at King Hussein Medical Center at the Royal Medical Services, Amman, Jordan from January to December 2009. The study design involved an open label retrospective analysis of medical records of 2 groups of patients of comparable age and onset of first stroke that received injections of botulinum toxin into the calf muscles as part of a rehabilitation program. Inclusion criteria were patients of any age and either gender who were able to mobilise aided or unaided at the physiotherapy department, and were clinically suitable for a combined BTX-A treatment and physiotherapy. Patients were excluded if they had fixed contractures in their lower limbs, hypersensitivity to any of the injected material, or had significant changes in their oral anti-spastic medications during the follow-up period. All patients chosen for the retrospective study were able to walk at least 10 meters with an appropriate walking aid and/or orthosis (if required). The purpose of the injection was mainly to provide a therapeutic opportunity for physical therapy to improve gait, balance, and for pain control. The BTX-A (Dysport®, Ipsen Ltd, Slough, Berkshire, UK) was presented as a freeze-dried pellet of 500 units of botulinum A toxin-hemagglutinin complex, 125 mg of human albumin, and 2.5 mg of lactose. The contents of the vial were reconstituted in one ml of 0.9% sodium chloride solution. Group I received Dysport® 500 mU into the lateral and medial heads of the gastrocnemius muscle. Group II received the same dose delivered to 4 sites, including the soleus (Figure 1). The injection was carried out using a one ml syringe with 25-gauge needle. Muscle identification was carried out visually and by palpation due to the obvious size of these muscles. The total dose was divided equally between injection sites. How long it took the patient to achieve the ability to walk 10 meters (in seconds) was the single outcome measure used to evaluate the functional gain as measured immediately following the injections (M0), and at the end of month (M) one, 3, and 6. The Modified Ashworth Scale, the presence of pain, and the use of orthoses or walking aids were noted but not included in the analysis. All patients received physiotherapy by the same team, which included passive and active exercises. Ethical approval was not deemed necessary as the study was merely extraction of data from medical records; however, permission was obtained from the Institution’s Higher Management Board. The study was performed in accordance with ethical standards laid down by the Declaration of Helsinki in 1964.
Illustration of the injection sites of the gastrocnemius muscle with an alternative injection technique. Group I: Two injection sites. Group II: Four injection sites.
Statistical analysis
Statistical analysis was carried out using the Statistical Package for Social Sciences (SPSS Inc., Chicago, IL, USA) version 16. Continuous data were presented as mean (± SD). Confidence intervals at 95% (CI) were calculated where relevant. Student’s t-test was used to compare paired data within the same group. An analysis of differences between means was used to examine non-paired data.
Results
There were no significant differences between the 2 groups in terms of gender, age, duration, and side of hemiplegia (Table 1), and the cause of stroke. In group I, the mean (±SD) walking time at M0 was 34 (±8.6) seconds (CI: 28.3-39.7), which was significantly different to that at M1, which was mean 23 (±6) seconds (CI: 19.2-26.8; p=0.0057). At M3 the mean was 22 (±7) seconds (CI: 17.6-26.5; p=0.0053 compared with M0), but not different than that at M6, mean 25 (±7) seconds (CI: 20.6-29.5; p=0.059 compared with M0). In group II, the mean (±SD) walking time at M0 was 36 (±12) seconds (CI: 29.1-42.9), which was significantly different than at M1, at mean 24 (±9) seconds (CI: 18.8-29.2; p=0.012); at M3 mean was 24 (±8) seconds (CI: 19.4-28.6; p=0.015 compared with M0), and at M6 the mean was 26 (±9) seconds (CI: 20.8-31.2; p=0.035 compared with M0). However, the analysis of the difference between the means showed no significant difference between the 2 groups in the 10-meter walking time at 1, 3 and 6 months in both group I and II (Table 1).
Comparison of spastic equinovarus deformity patients’ characteristics in Group I (2 injection sites) and Group II (4 injection sites).
Discussion
Both the lateral and medial heads of the gastrocnemius muscle are responsible for plantar flexion of the foot with the knee extended, while the soleus muscle is responsible for plantar flexion of the foot with the knee flexed.9,12 Foley et al14 carried out a systematic review and meta-analysis of the literature and found that the use of botulinum toxin in the management of lower limb spasticity after stroke produced a small, but statistically significant increase in gait velocity. In a double-blind, placebo-controlled, dose-ranging study, 234 patients with hemiparesis with spastic equinovarus deformity of the ankle after stroke were randomized to one of 4 treatment groups: 500 units of Dysport®; 1000 units of Dysport®; 1500 units of Dysport®, and placebo. Patients were assessed every 4 weeks over a 12-week period. The distance covered during a 2-minute walking test was significantly increased in each these groups, but there were no differences between groups. Significant improvement in calf spasticity, limb pain, and reduction in use of walking aids was noted in the Dysport® groups relative to the control group.15 Rousseaux et al16 demonstrated BTX-A to reduce the degree of spasticity; however, the effects on gait and activities of daily living remained controversial. In a series of 47 stroke patients treated with BTX-A for spastic leg muscles, they showed a moderate but significant reduction in spasticity and a slight increase in gait velocity and step length, with great variability among patients. Isoyama and Takeuchi17 provide an overview of BTX-A as a treatment option for spasticity in stroke patients. They particularly reviewed the combined effects of BTX-A injection into spastic muscles with physiotherapy modalities. Other studies showed that BTX-A when combined with physiotherapy or casting may be useful for improving motor function in stroke-induced spasticity.18 This study demonstrated no significant difference between 2 comparable patient groups who received similar management measures except for the number of injection sites of BTX-A (Dysport®). The rehabilitation program employed the principle of early use of botulinum toxin to reduce the muscle tone of the calf muscles.9 All patients in this study were treated on an outpatient basis after completing a standard structured inpatient rehabilitation program. They were referred from a single Neuroscience center, and received rehabilitation intervention by the same personnel using a standard neuro-rehabilitation approach.18
In this retrospective study, it was noted in group II that the beneficial effect of BTX-A was still present at M6 compared with baseline. This effect was only seen up to the M3 in group I, which received the toxin in 2 sites. The chosen dose of Dysport® (500 mU) was considered very modest for the size of the calf muscles.15 Similar to previous reports, this dose appeared to be sufficient in reducing the tone to that the point it improved pain perception,19 gait,20 and facilitated the use of ankle/foot orthosis.21-23
The value of reducing the number of injection sites would help in minimizing patient discomfort, and possibly the production of antibodies.12,18 Further research in larger samples using randomized controlled studies is required to establish the value of injecting the soleus in addition to the gastrocnemius muscle especially after stroke. In addition, other factors may need to be considered and taken into account.
Footnotes
Disclosure
Trial medications were made available on purely clinical grounds. No commercial or pharmaceutical companies had any role in study design, collection, analysis, interpretation of data, in the writing of the report, and in the decision to submit the paper for publication. Imad M. Al-Khawaja received travel grants from Allergan Inc., and Ipsen Ltd.
- Received February 5, 2014.
- Accepted May 6, 2014.
- Copyright: © Neurosciences
Neurosciences is an Open Access journal and articles published are distributed under the terms of the Creative Commons Attribution-NonCommercial License (CC BY-NC). Readers may copy, distribute, and display the work for non-commercial purposes with the proper citation of the original work.